Pick up a dense clump of ordinary office staples and pull. It won’t budge. It feels like a solid block of steel, rigid and stubborn, resisting every Newon of force you throw at it. But give it the right kind of shake—a specific, calibrated vibration—and the entire structure collapses, flowing through your fingers like a stream of silver water. This isn’t a magic trick, and it isn’t a chemical reaction. It is a glimpse into a future where our buildings, robots, and bridges are made of materials that can decide whether to be a brick or a puddle on command.
Traditional materials are boringly predictable. If you have a pile of sand, the grains are convex—they are smooth, rounded, and essentially selfish. One grain of sand doesn't care what the next one is doing; they just slide past each other. This is why you can’t build a vertical wall of dry sand. It has no “tensile strength,” meaning it can’t hold itself together when you pull on it. To make a solid structure out of small parts, we usually have to add a “glue,” like the water in a sandcastle or the cement in concrete. But the Barthelat Lab wanted to ditch the glue entirely.
The end of the convex grain
When you dump these staple-shaped particles into a pile, they don't just sit there. They weave. Much like the sticks in a bird’s nest or the fibers in a wool jumper, the legs of one particle snag the body of another. This creates a collective strength that is greater than the sum of its parts. In “pickup tests,” the researchers found that a tangled mass of these staples could be lifted as a single, rigid unit. It behaves like a solid because the physical geometry prevents the particles from sliding. You are essentially building a material where the “bonds” are mechanical rather than chemical.
This creates a rare physical paradox: a material that is both strong and tough. In materials science, these two terms are often at odds. A ceramic plate is strong (it can hold a lot of weight) but it isn’t tough (hit it with a hammer and it shatters). A rubber band is tough (it absorbs energy and stretches) but it isn’t strong. These entangled staples somehow manage to do both. They resist being pulled apart with the stubbornness of metal, but because they can slightly shift and slide against each other, they absorb impacts without breaking. It is the kind of mechanical property that makes engineers salivate.
How do you turn a bridge into a puddle?
The real genius of the CU Boulder research isn't just that the staples stick together—it’s that they know when to let go. This is the “liquid metal” aspect that has everyone from NASA to the Ministry of Defence interested. By applying specific patterns of vibration, the researchers can effectively “turn off” the material’s solidity. Gentle vibrations actually help the particles find their groove and interlock more tightly, making the structure stronger. But hit the “release” frequency, and the particles are jostled out of their mechanical embrace. The “solid” block suddenly liquefies, allowing the staples to be poured into a new mold or cleared away entirely.
This raises a fascinating set of questions for the future of construction. Imagine a temporary bridge deployed in a disaster zone. Instead of heavy steel beams that require massive cranes and permanent bolts, you could pour a “slurry” of these interlocking particles into a frame, vibrate them until they lock solid, and walk across. When the job is done, you hit the shake button, the bridge melts back into a pile of staples, and you shovel them into a truck to use again at the next site. It is the ultimate recyclable infrastructure.
However, there is a catch. Using mechanical entanglement on a large scale is currently expensive and incredibly difficult to simulate perfectly. While the computer models can predict how a thousand staples will behave, predicting how a billion of them will react under the weight of a moving lorry is a different beast entirely. There is also the issue of “fatigue.” If the material relies on tiny metal legs hooking together, what happens when those legs start to bend or snap after the thousandth “melt” cycle? The team is already looking at more advanced designs, including particles with even more legs—similar to the spiky burrs that stick to your dog’s fur—to create a more permanent, fail-safe lock.
Nature’s messy rulebook is being rewritten
The CU Boulder staples are part of a broader, weirder trend in physics: we are realizing that the simple “solid, liquid, gas” diagrams we learned in school are mostly lies. While Barthelat is playing with macro-scale staples, other physicists are finding the same “strange” behavior at the atomic level. Recent studies into gallium—the metal that famously melts in your hand—have shown that its liquid state is far more structured and “solid-like” than we ever suspected. Gallium doesn't just turn into a random soup of atoms; it retains a ghostly memory of its crystal structure even while it’s flowing.
Further down the rabbit hole, researchers working with graphene and superfluids have observed a “quantum pinball” state. In these systems, electrons can be made to “freeze” into patterns that look like solids but behave like liquids, or vice versa. We are entering an era where the “phase” of a material is no longer a fixed identity, but a temporary mood. Whether it's a bunch of office staples or a sheet of atoms, the rules are the same: if you can control the geometry and the energy, you can make matter do whatever you want.
This brings us back to the T-1000. While we aren't quite at the stage of building a shapeshifting assassin that can imitate your step-mum, the CU Boulder team believes the immediate future lies in swarm robotics. Imagine a thousand small, simple robots—none of them smarter than a toaster—that can hook into each other to form a solid tool, like a wrench or a ladder. When the task is done, they disentangle and flow into a small box for storage. It is a transition from “hard” robotics to “soft” systems that can adapt to their environment.
The cost of the shapeshifter
But why isn't this already in our homes? The barrier, as always, is the messy reality of the physical world. For a material to be truly useful, it has to be reliable. If you build a chair out of these staples, you need to be 100% sure that a passing heavy lorry or a loud bass speaker won't hit the “melt frequency” and leave you sitting on the floor in a pile of metal. Engineering the “off switch” to be specific enough that it won't be triggered by accident is the next great hurdle.
There is also a regulatory nightmare waiting in the wings. Our current safety standards for buildings and machines are based on the idea that solids stay solid. How do you certify a bridge that is designed to fall apart? How do you write a building code for a structure that could, in theory, be poured into a drain? These are the types of friction points where cutting-edge physics meets old-world bureaucracy.
Despite these hurdles, the momentum is undeniable. We are moving away from a world where we build things out of static blocks and toward a world where materials are active participants in their own function. The humble office staple, a tool we usually use to keep a few sheets of paper together, has inadvertently shown us the blueprint for a more fluid future. It turns out that if you want to change the world, you don't necessarily need a new element or a revolutionary chemical—you just need a better way to get things tangled up.
The next time you drop a box of staples on the floor and spend ten minutes trying to untangle them, don't get angry. You’re not looking at a mess. You’re looking at the strongest, toughest, most adaptable building material we’ve ever conceived. You just haven’t found the right frequency to make it behave yet.
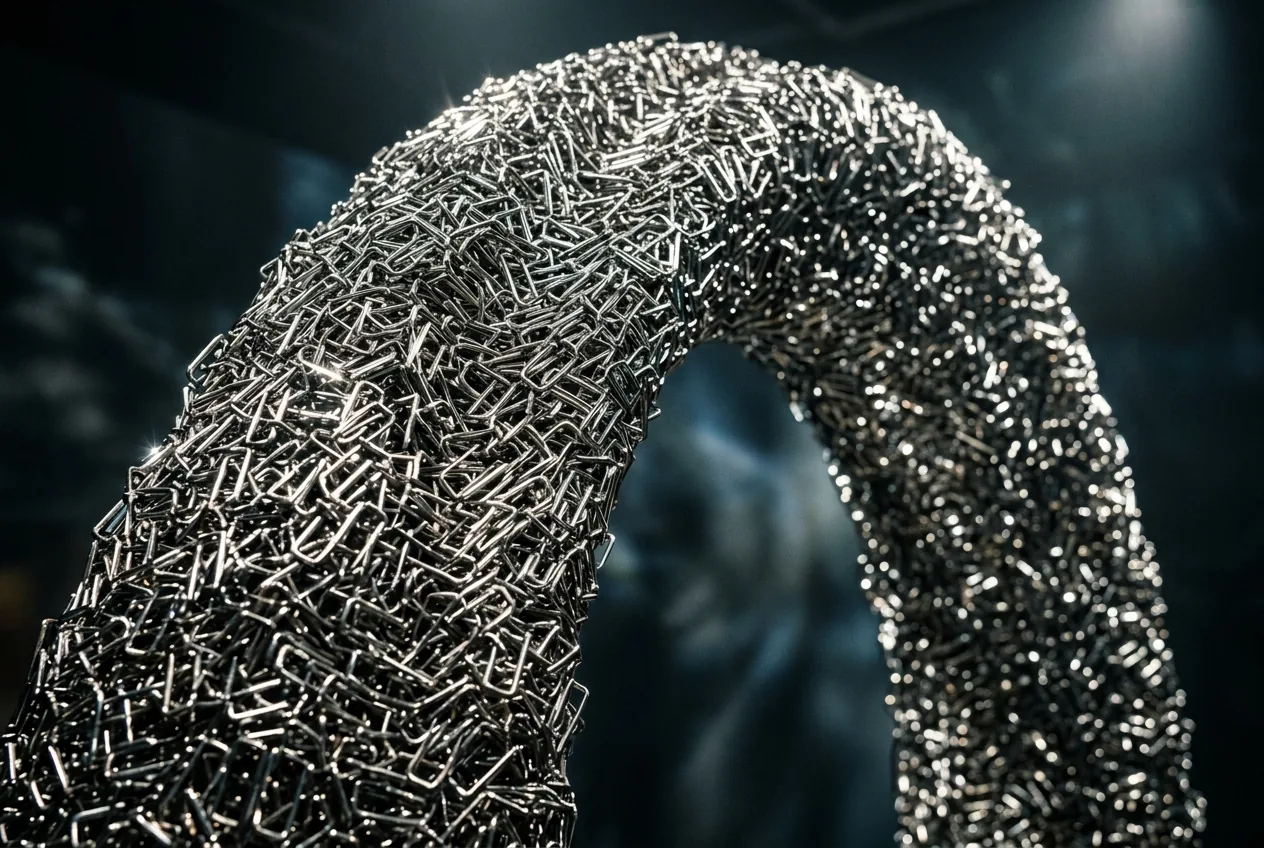
Comments
No comments yet. Be the first!