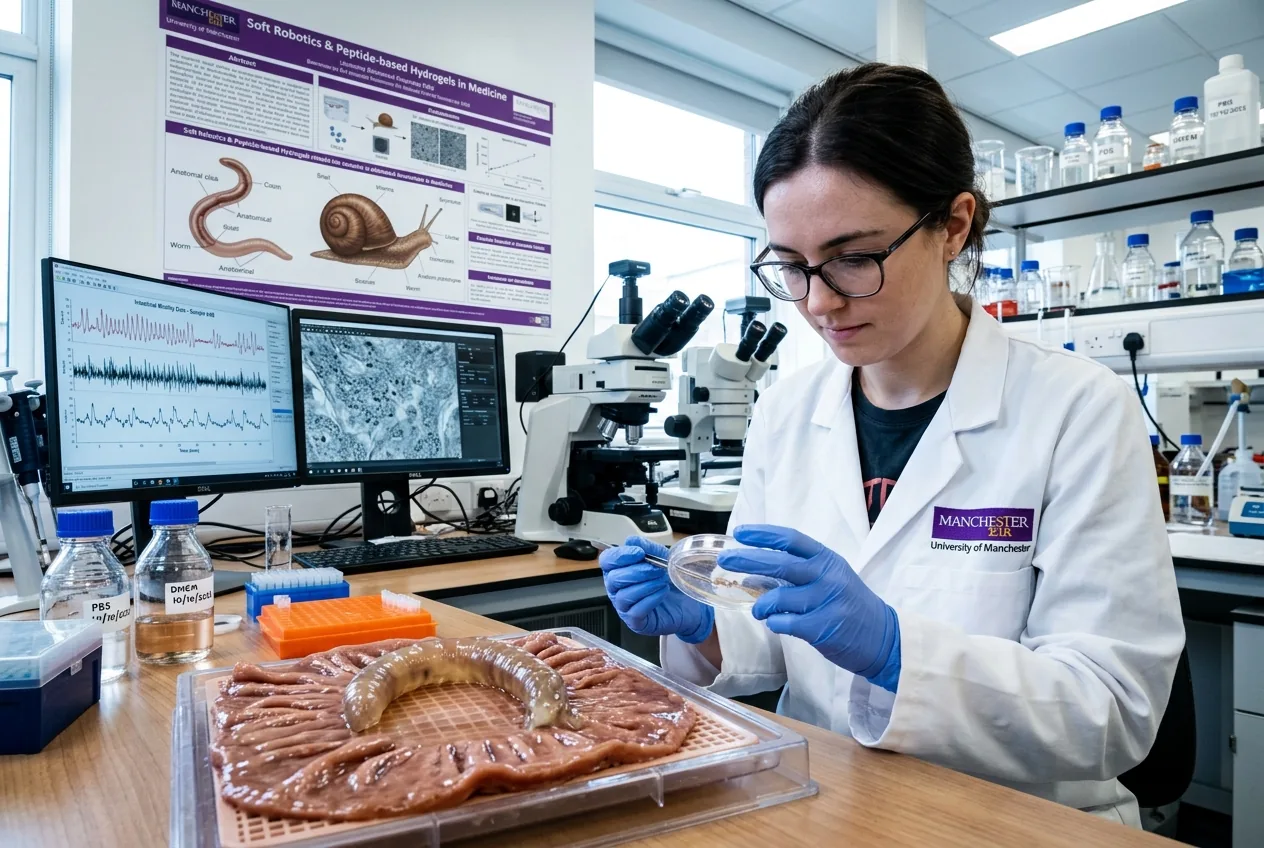
Manchester's lab got an unusual brief: study snails and build tiny robots that can sit on a tumour and release medicine.
Under fluorescent lamps and beside microscopes, Dr Mostafa Nabawy's group at the University of Manchester has been granted nearly £1 million from UK Research and Innovation to do what sounds almost mischievous: learn how snails move and then copy that behaviour inside the human gut. At the heart of the project is a simple, counterintuitive idea — snail-inspired robots use slime-like motion and rhythmic waves to adhere to slippery, uneven surfaces and, critically, to anchor and deliver drugs precisely to bowel tumours. The initial funding pays for high-resolution datasets, peptide-based bionanomaterials and the digital-twin software needed to test designs in silico before anything touches a patient.
The nut: why this matters now
Colorectal cancer remains one of Europe's most common cancers, and systemic chemotherapy — the blunt instrument still widely used — causes dose-limiting side effects because drugs circulate through healthy tissue. If tiny devices could deposit a therapeutic payload directly at a tumour and release it controllably, clinicians could increase local drug concentration while reducing systemic toxicity. The timing is also tactical: soft robotics, advanced biomaterials and machine-learning simulation tools are maturing simultaneously, creating an opportunity to attempt a genuinely new class of intrabody devices rather than incremental improvements to pills or catheters.
How snail-inspired robots use slime-like motion to anchor and steer
Biologists and roboticists have long admired the snail's repertoire: slow travelling waves along a muscle-lined foot, coupled to a thin adhesive mucus layer, let a snail crawl across stones, glass and vegetation without cutting the surface. The Manchester team is translating those mechanics into soft robotic actuators that produce travelling deformations and secrete, or emulate, a thin lubricating/adhesive layer. In practice this means a robot can switch between low-friction sliding for transit and high-adhesion anchoring for drug release, a capability that existing capsule endoscopes or microswimmers struggle to offer. That switching is the core advantage for precise cancer drug delivery: the device can position itself close to malignant tissue, latch on without puncturing healthy mucosa, and then dose locally over time.
Designing controllable systems: how snail-inspired robots use slime-like locomotion and digital twins
Control is where the project departs from classroom biology. The robots are planned to be made from peptide-based bionanomaterials that can be tuned at the molecular level and respond to benign external triggers such as magnetic fields. To avoid blind trial-and-error inside the lab or, worse, a patient, the team will build a multiscale digital twin: a simulation stack that couples biomechanics, mucus rheology, robotic actuation and tumour mechanics. High-resolution experimental datasets on real snail foot actuation and mucus interactions will train machine-learning models to predict how a specific gait will behave on human mucosa. The digital twin compresses what would be years of benchwork into a virtual design loop — but it also creates a dependency on accurate, generalisable data and on validation against living tissue models.
Peptide bionanomaterials, magnets and the trade-offs of 'soft' design
Peptide-based materials promise biocompatibility and chemical tunability: you can design a polymer to soften at body temperature, to degrade after a preset interval, or to bind selectively with a target. Coupling those materials with embedded magnetic particles allows remote nudging and orientation from outside the body. That sounds elegant, but it introduces trade-offs. High magnetic susceptibility improves controllability but raises concerns about imaging interference and heating under alternating fields. Likewise, soft structures are excellent at conforming to tissue but complicate sterilisation, batch-to-batch manufacturing and long-term mechanical reliability. For engineers, the question is never whether a trick works in the lab; it is whether it scales and passes toxicology and regulatory guardrails without turning into a hobbyist's artefact.
Clinical and translational hurdles: imaging, safety and the gut as an adversary
The gastrointestinal tract is a hostile environment for precise robotics. Mucus thickness, pH, peristaltic motion and the microbiome vary between patients and even along the bowel of the same patient. A device that sticks in one patient could wash away in another, or worse, block a lumen. Real-time localization is another Achilles heel: steering via magnetic fields requires an independent way to see where the robot actually is. Conventional MRI is incompatible with many magnetic actuation schemes; X‑ray or fluoroscopy expose patients to ionising radiation. The project’s digital-twin approach mitigates some risks by simulating robot–tissue interactions, but preclinical validation — organ-on-chip, ex vivo mucosa and animal models — will still be lengthy and expensive. Expect a timeline measured in years, not months, before first-in-human trials can even be considered.
Advantages, unanswered problems and the role of machine learning
There are clear technical advantages to slime-like locomotion for targeted therapy. Adhesive locomotion lets a robotic device maintain a precise spatial relationship to a tumour while delivering repeated, localised doses — potentially unlocking dose schedules that systemic chemotherapy cannot achieve. Machine learning helps by turning messy experimental observations of snail gait and mucus rheology into controllers that can adapt to variation in the wild. However, ML models are only as robust as the data they are trained on; if initial datasets do not capture patient diversity — different ages, disease states, mucus chemistries — controllers may fail in the clinic. Robustness, interpretability and safety-guarded control policies will be as important as the materials and magnets.
Europe, funding and the politics of medical robotics
The project highlights broader industrial-policy questions. The grant comes from UK Research and Innovation’s cross-council responsive mode, a flexible fund aimed at interdisciplinary projects, and shows the UK is still investing in high-risk, high-reward bioengineering despite leaving the EU. On the continent, research funding and device regulation follow different rhythms: EU programmes favour large consortia and longer lead times, while national quick-response funds can move faster but at smaller scale. Manufacturing medical-grade peptide materials at scale will need supply chains that stretch across Europe — peptide synthesis, GMP facilities, sterilisation experts — and those chains are unevenly distributed. In plain terms: Manchester can build prototypes, but scaling to a commercial medical device will require coordination with EU regulatory pathways, clinical partners and manufacturing sites that may sit in Germany, the Netherlands or beyond.
How close are snail-inspired systems to clinical use?
The short answer is: not imminent. The current funding supports early-stage development: experimental datasets, material chemistry, proof-of-concept actuators and digital-twin simulation. Those are necessary foundations but far from clinical validation. Translational steps — GLP toxicology studies, reproducible manufacturing methods, imaging-and-control integration and regulatory submissions — will take several more funding rounds and years of work. Researchers are candid about this: the aim now is a platform that could transform delivery paradigms, not a product ready for the hospital shelf next year.
In the end, Manchester has the biology and the clever engineers; Brussels and Berlin will have to decide where the factories and clinical pathways go. Germany has the machinery; Brussels has the paperwork; Manchester has the snail — and now it needs the money and a lot of patience.
Sources
- The University of Manchester (project and press materials)
- UK Research and Innovation (CRCRM funding scheme)
- Nature Communications (2024 study on sliding motions and soft robotic locomotion)
Comments
No comments yet. Be the first!