A child answering the kettle: the small moment that reframed a headline
She answered her mother from the kitchen sink: not a clinical milestone, but the sort of ordinary scene parents keep in their pockets. Four months after a single operation, the seven‑year‑old could follow a conversation in the home — she could hear the kettle. That image has been repeatedly recycled by press materials and headlines, and it helped fuel the phrase 'deafness reversed': scientists restore hearing in 10 patients after one injection. It is an accurate description of what the trial reported, and it is also where the simple story stops being sufficient.
'deafness reversed': scientists restore — the numbers that make headlines
The trial enrolled ten participants aged 1 to 24. Across the cohort the average level of sound the patients could perceive dropped from about 106 decibels to 52 decibels within six months — a shift that converts extreme hearing loss into a range where conversational speech is possible for many people. Improvements were often rapid: several patients began to show function after just one month, and the youngest children — particularly those aged five to eight — had the largest gains. One seven‑year‑old regained almost full functional hearing and was able to speak on the phone and carry daily conversations with family.
Those figures explain why the press ran the words 'deafness reversed' and why parents and clinicians are excited. They also contain an obvious statistical caveat: ten people is still a small sample. The study is a single‑arm trial with no randomised control, and follow‑up so far is limited to months rather than years. The result is a powerful demonstration of biological plausibility for this specific mutation, but not yet a universal cure.
'deafness reversed': scientists restore — limited to OTOF, and that matters
The patients treated all shared the same molecular diagnosis: biallelic loss‑of‑function mutations in OTOF, the gene encoding otoferlin, a protein critical for the synaptic transfer of sound information from inner hair cells to the auditory nerve. Delivering a correct copy of OTOF to those hair cells restored that communication and produced the measurable improvements reported.
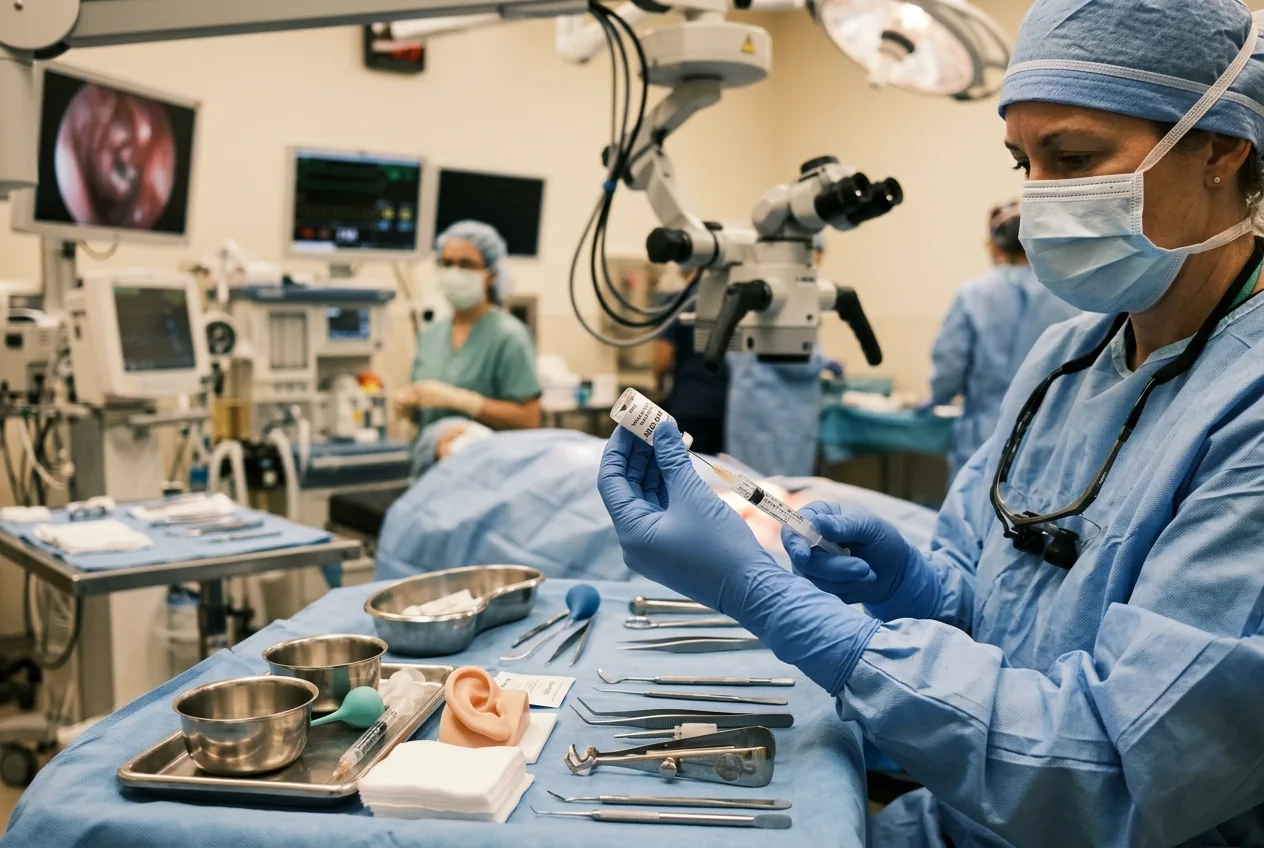
The operative moment and the biology reported in the paper
The procedure in the trial was surgical and local: clinicians injected an AAV‑based vector carrying a functional OTOF sequence through the round window membrane into the cochlea. The inner ear is a confined, delicate space; the operation requires otologic surgical skill and imaging. The trial teams report that the intervention was generally well tolerated. The most common laboratory change was a temporary reduction in neutrophil counts and, crucially, no serious adverse events were reported during the six‑ to 12‑month follow‑up window.
That safety profile will need to be watched closely. Adeno‑associated viral vectors are widely used in gene therapies because they are comparatively benign, but the cochlea’s small volume and the proximity of treated cells to the central nervous system raise specific questions about dose, immune response and long‑term expression. Neither the trial nor the paper can yet answer how durable the effect will be over years or decades, whether repeated dosing will be needed, or how bilateral (both ears) treatment performs compared with unilateral dosing.
Commercial ties, conflicts and the race to treat common deafness
Beyond conflicts, there is a more systemic tension. Each genetic form of deafness will need its own tailored treatment: new vectors, new manufacturing, bespoke surgical protocols and fresh safety dossiers. That bespoke nature is why many groups — including Rinri Therapeutics at the University of Sheffield — are pursuing a different tack, developing stem cell‑derived auditory nerve progenitors aimed at a wider class of sensorineural losses. Cell therapies promise broader applicability, but they bring their own regulatory, manufacturing and delivery hurdles. The economics are stark: personalised gene medicines and complex cell products are expensive to develop and scale. Who will pay, and how quickly will regulators accept the new approaches, remain open questions.
Is the treatment available now — and what are the real risks?
Short answer: no, not for routine clinical use. The Nature Medicine trial is an early clinical step. The product remains experimental, and the pathway to wider approval involves larger multicentre trials that demonstrate durable benefit, acceptable safety across diverse populations, reproducible manufacturing, and clarity about cost and access. For families hearing the headlines, that can be an uncomfortable message: the science is real, and the therapy worked for these patients, but availability to the public will take additional trials and regulatory review.
As for risks, the published follow‑up showed no serious adverse reactions in the first year, but clinicians and regulators will watch for late immune effects, off‑target expression, and the unknowns of long‑term cochlear biology after genetic restoration. There is also the practical surgical risk of accessing the cochlea and the possibility that patients who receive the treatment at older ages will gain less than children because of long‑standing neural deprivation.
Where this sits in the longer arc toward restoring hearing for millions
The trial is an inflection point: it proves a gene therapy can be delivered safely into the human cochlea and produce rapid, meaningful gains for a genetic subgroup. That is rare and important. But the history of medicine is littered with early miracles that proved difficult to generalise. The next steps will be pragmatic, expensive and slow: scaling manufacturing, running randomised trials, securing regulator approval in multiple jurisdictions, and building surgical and audiological systems to deliver and monitor a new class of treatments.
Meanwhile, complementary avenues such as Rinri’s Rincell‑1 neuron replacement programme aim at the larger market of sensorineural loss from ageing and noise damage. Those technologies, if they succeed, could reach more people but will face their own proof‑of‑concept and delivery hurdles. The immediate lesson is that the field is now legitimately bifurcated: targeted gene fixes for specific monogenic deafness, and broader regenerative approaches for acquired or complex forms.
The kettle in a kitchen is still a good way to measure meaning. For the families of the trial participants a single operation produced life‑altering changes within months. For the rest of the world the headline 'deafness reversed': scientists restore is a promise in progress rather than a finished story. The careful work now is on durability, scale, equity and independent replication; the very practical question is whether regulators and health systems can convert an elegant, gene‑specific success into fair, lasting access for those who need it.
Sources
- Nature Medicine (AAV gene therapy for autosomal recessive deafness 9: a single‑arm trial)
- Karolinska Institutet (press materials and institutional research communications)
- Zhongda Hospital, Southeast University (clinical collaborators)
- Rinri Therapeutics / University of Sheffield (Rincell‑1 and regenerative cell therapy programme)
- Otovia Therapeutics (developer and funder involved in the trial)
Comments
No comments yet. Be the first!